Also, the S5 starts blinking, then shuts down at somewhere around 3v.
Honestly any quality 18650 will be fine. You will notice the massive drop in output before you over discharge the li ion. Anybody who runs their protected cells to cut off is already doing it wrong. With an in protected once high stops being much brighter than medium, time to charge.
Protection is only a back up. It isn’t a magical fix to make the battery never explode. All it does is protect the battery from chargers that over charge, users who never re charge their batteries, and shorts. I prefer unprotected because the protection circuit hurts battery performance.
This. Having actually done many run time tests on lights where I leave them on and just let them go, most lights have a way of telling you it’s time to charge them. They get really dim. Constant output drivers are found on nicer lights anyway. And the thing is they get noticeable dim for quite some time before they ever reach 2.7-2.8ish. I run some laptop pulls in a convoy s6 and since the light isn’t used a whole lot, I charge it once every two or three months. It’s never anywhere near 3.7 volts. Most lights begin to dim after 3.7 volts…. so you have an entire volt of dim light that acts as an idiot warning. That volt is actually quite a long time of noticeably dim output.
most lights have a way of telling you it’s time to charge them. They get really dim.
+1 for Dimbo, but I must point out that there are a lot of people on this forum who won’t notice the difference between High and Medium (the first “needs charging” indicator) because they never use High or Medium!
However, comma, I have recycled more bad laptop 18650s than what good ones I have for use now, and those reached ~0VDC without any “undervoltage” incidents. In fact, the only way I have to find them is the VOM, or stick them in a flashlight. It’s strange watching one “self-discharge” from ~4v to ~0v in your hand. So much for that end of the spectrum.
No overvoltage capability, with the single- and double- chargers I got “for free” (replacement cost for both ~3 bucks), I tested them for many recharges outside on the porch. I finally quit leaving a “ready spare” on the charger because between whatever’s in the light when I pick it up, plus the “pocket spare” 18650 I carry with me, plus the few recently-charged ones lying about, I just got tired of looking at the green LED.
I would honestly like to see the “worst-case scenario” with an 18650. I have actually blown up a 12VDC SLA car battery at about solar-plexus level, which scattered parts across Main St. Columbia, SC and sent me inside to wash the electrolyte off my face & I had to throw away the “business-casual” shirt and tie I was wearing. Do as I say, not as I do! But the event was only annoying, not actually dangerous, except for the scary face splash. Obviously I blinked quickly enough (plus sunglasses) that none got in my eyes!!
I’m not doubting that the Lithium chemistry is scary and exciting, but I suspect a little bit of “believing” in the horror stories…
So my side project for now is to rig a way to make a dead short across a fresh-charged 18650 (NOT at solar-plexus level!!) so I can “know” what to expect.
I’ll use my smallest SD card for the video. If it fills up before anything bad happens, “I’m calling this myth ‘busted’”… Sorry…
(There’s a method to this madness, you know…
Where I work sometimes, Safety is more important than profitability or productivity. “Intrinsically Safe” means there’s nothing in the item which could cause a fire, regardless. They’re stuck with 2*AA Streamlights now, and my “pocket spare” 18650 gets some nasty looks. (Yes, some of those “pocket spares” came from “dead” laptop packs from that same plant…) A 4v spark would condemn the Lithiums to the parking lot. If the first test isn’t too embarrassing, I’ll try one in a stoichiometric mix of Propane and air… This could be fun… BRB…)
I found the story I read on CPF:
http://webcache.googleusercontent.com/search?q=cache:HDHTUWdZrD4J:www.candlepowerforums.com/vb/showthread.php%3F141137-Inhaled-vapors-from-battery!!!
I found the story I read on CPF:
http://webcache.googleusercontent.com/search?q=cache:HDHTUWdZrD4J:www.candlepowerforums.com/vb/showthread.php%3F141137-Inhaled-vapors-from-battery!!\!
That venting was with a primary lithium cell CR123, which according to the “Gods” at the other forum are more dangerous ![]()
See excerpt from greenbatteries.com
“There are several important differences. The practical difference between Lithium batteries and Lithium-ion (Li-ion) batteries is that most Lithium batteries are not rechargeable but Li-ion batteries are rechargeable. From a chemical standpoint Lithium batteries use lithium in its pure metallic form. Li-ion batteries use lithium compounds which are much more stable than the elemental lithium used in lithium batteries. A lithium battery should never be recharged while lithium-ion batteries are designed to be recharged hundreds of times”
First of all, I think you are fully qualified to use Li-ions. Remember to respect them, but don't be too afraid.
Oh, the Convoy S5 has a firm clicky so I am confident it will not accidentally turn on when it’s in my coat or handbag.
Especially after scaru's incident, I'm not confident with any light not accidentally turning on.
Luckily the S5 has anodized threads and it can be locked out by opening the tailcap about 1/3 turn. That's what I'm always doing with mine when putting it to backpack (or similar).
FWIW, I use unprotected flat top NCR18650B in my S5. Fits well & offers good capacity.
Over voltage protection is provided by the charger and under voltage protection by S5 driver. Possible dangerous event would be the light shorting during use, but then the user can notice & act on that. (If the light turns off for any reason, open the tail cap and remove cell.)
That venting was with a primary lithium cell CR123, which according to the “Gods” at the other forum are more dangerous
Wow, really? I did find a couple of explosion threads featuring that cell but who knows if they were mixing cells of different brands etc.
There was this thread where a cell exploded whilst inside a light and injured the poor owner. 3x18650 but he didn’t measure the cells before using them. :~
First of all, I think you are fully qualified to use Li-ions. Remember to respect them, but don’t be too afraid.
I remember reading all these disastery threads before I joined BLF and they can put you off especially as I didn’t really know. Even now after rereading them, I am wondering if I should really leave a cell inside a light I use often. “What if it vents?” “Will the light explode?” ![]()
Yea, there is always a small risk but the chances should be low for you at least. Your looking for good cells & you won’t abuse them.
You can start out (or stick with) Li-Mn / IMR.
Did you go for those AW IMR 2000mAh 18650s? Any li-ions on the way?
I didn’t get any AW yet. I thought they were out of stock but I noticed two AW 2000mAh is 26 USD shipped, compared to 14.79 USD for a pair of Panasonic 3400mAh.
Also, 2000mAh doesn’t sound too low but I was also planning on using the ML-102 with a cell to top up my phone. I’m not really sure how much it needs to be.
I found the story I read on CPF:
Vapors aren’t the real problem in a chemical factory (being more of a “normal course of business” kind of deal), but that’s darned interesting.
It’s amazing what you can observe, just by looking!
Not tending to trust blogs and chatroom posts (present company EXcluded, of course!), I went and found something only mildly interesting if directly relevant:
GCMS Analysis of Gas Generated from Lithium Ion Rechargeable Battery Cells
You’ll notice there’s no HF, and there are those unlabelled peaks… Not sure what to make of this, especially with his 80C temperature limit, but it is directly relevant.
But let’s look at the CPF story itself. When I was maybe 10, we found a discarded gallon bottle of H2SO4. Yes, I sniffed it. Do NOT try this at home!! But, it seems to have somehow enhanced my sense of smell. All ends well in Dimbo’s weird world! So this story pushes a couple of my buttons. But wait…
HF, sniffed or even touched, is exactly as bad as the writer implies!!! His claim of “last night” isn’t plausible, given the rest of the story, but a cautionary tale isn’t told in court. So don’t let me diminish his advice. Think! Pay Attention! Be Very Careful With Chemicals! Even DiHydrogen Monoxide will kill you quicker than keep you alive!
But WAIT…
For DECADES now, in “the land of the Free, the home of the Brave”, “we the People” have allowed anonymous agents of our government to add Flouride to our water supply. You can say that softer, less scary, but it’s no less a fact.
If you track down the HF lead, guess what the MSDS says? No, really you must guess or I will pout!!
Keep your sense of humor…
Here’s some Truth, to still Laughter in you forever:
Hydrofluoric Acid Chemical Safety Information
If you’re not going to read the whole thing, before you drink public water again, at least read the last page:
Fluoride from any source has the same toxicity. A mass poisoning occurred at a state hospital many years ago when a bug poison containing NaF was mistaken for powdered milk and added to scrambled
eggs. There were reported to be 47 deaths. In some regions of the country the levels of fluoride in the
water are too high and this causes mottling of the teeth and loss of calcium from the bones.
Inhalation of HF produces an immediate injury to
the lining of the lungs with hemorrhage pulmonary
edema and death. It may take only about 5 minutes of
exposure to inhaled HF to produce death in a
couple of hours.
All in all, fluoride from whatever source can be very dangerous.
Ask your nearest bureaucrat, how do they put Sodium Fluoride (NaF) in the water they make you pay to drink? They’ll bury you in BS, but you can beat them back with the Material Safety Data Sheet for Sodium Fluoride . Especially this section:
Special Remarks on other Toxic Effects on Humans:
Acute Potential Health Effects: Skin: Causes skin irritation and possible burns, especially if skin is wet or moist. Eyes: Causes eye irritation and burns. May cause chemical conjunctivitis and corneal damage. Ingestion: Harmful if swallowed. Causes digestive (gastrointestinal) tract irritation and burns. May cause severe and permanent damage to the digestive. Ingestion of large amounts may cause salivation, thirst, nausea, vomiting, hypermotility, diarrhea, and abdominal pain. May affect behavior/central nervous system/nervous system (headache, nervousness, dizziness, seizures, convulsions, tremor, muscle weakness, somnolence), respiration (respiratory depression, dyspnea), cardiovascular system (weak pulse, hypotension, dysrhythmias, cardiac arrest), liver, urinary system (polyuria, polydypsia) brain, metabolism (loss of appetite, hypcalcemia, hyperkalemia, hypomagnesia, ), teeth, bones, and blood (changes in red and white blood cell count, interference in blood coagulation) Inhalation: Causes irritation and chemical burns of the respiratory tract with coughing, breathing difficulty and possibly nasal
septum perforation and coma. May affect bones. Chronic Potential Heath Effects: Chronic ingestion may cause fluorosis. Effects of fluorisis may include joint pain, weakness, limited joint mobility, brittle bones, ossifications on x-ray, thickening of long bone cortices, calcificaiton of ligaments, osteomalacia, osteosclerosis (skeletal (bone and teeth) abnormalties) and mottled tooth enamel. Other symptoms may include anemia, nausea, vomiting, diarrhea or constipation, kidney damage and weight loss/anorexia. Chronic inhalation may cause bronchitis to develop with cough, phlegm, and/or shortness of breath, liver (hepatic enzymes increased, jaundice)
…
Yeah. You’re right. This doesn’t have a dad-gum thing to do with outgassing or any other Safety issue of 18650s.
But it could help explain the True Believers’ discomfort with Facts, Logic and Truth… “May affect behavior”…
At least it should bring some perspective to the CPF story…
Speaking WITH you all, of course, not ABOUT any of you!!!
Dim
(And no, I haven’t ginned up a dead-short test rig yet! All this put a bee in my bonnet about finding a willing chemist with a GCMS to test the result.)
That’s interesting! 80°C, you’d expect it to vent already! ![]()
I did find this, which might be biased? I don’t know what kind of Li-ion cells it refers to:
Furthermore, when the electrolyte is combined with water, there is the potential for hydrofluoric acid to form
But then this makes it sound like there needs to be a fire involved for it to be toxic:
One problem is that many lithium-ion batteries today contain fluorine, which readily combines with hydrogen to make hydrofluoric acid (HF). In accidental battery fires, HF is noxious, dangerous to the touch, and an inhalation danger.
PG/Duracell say in their MSDS:
Hazardous Combustion Products:
Thermal degradation may produce hazardous fumes of lithium and manganese;
hydrofluoric acid, oxides of carbon and sulfur and other toxic by-products.
But these are primary cells not rechargeables. :weary:
AW tends to charge a lot. You can get the same quality, often the same exact cell elsewhere for less.
Efest IMR 18650 2000mah outperforms AW IMR 2000mah on HKJ’s charts.
Panasonic CGR18650CH are kind of in between Li-mn / li-ion & are suppose to be good too… If the FT forum report of one venting hasn’t turned you off them. :face_with_monocle: ![]()
For li-ion I’d go for sanyo 2600mah over panasonic. The 3400mah sounds nice but it can’t keep its voltage up.
Scaru’s Li-ion Battery Safety and Shopping Guide with HKJ’s data.
Ahh, thank you! I hadn’t settled on the Panasonics. I need to do more reading. >.<
Aww, these are temporarily sold out and apparently discontinued (can’t find anything directly from Panasonic about this though):
http://www.fasttech.com/products/0/10001980/1141101-panasonic-cgr18650ch-rechargeable-2250mah-3-7v
I believe all panasonic "CGR" cells use an in-between chemistry. Panasonic seems to use "NCR" for their li-ion cells.
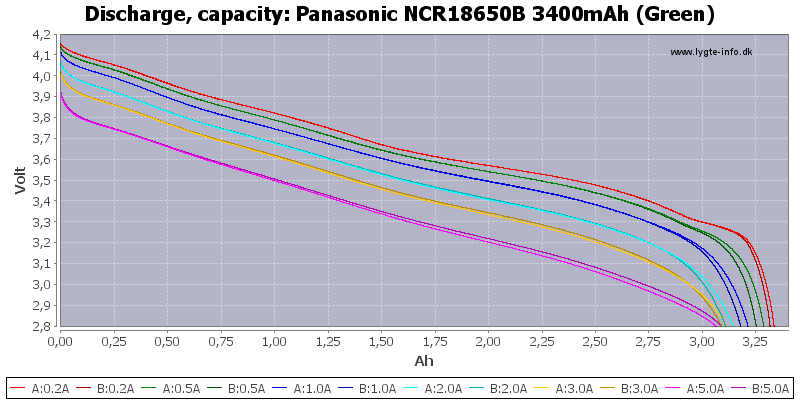
Panasonic is definitely high quality cells but our leds will dim before we can get 3400mah out of their NCR18650B. Even for charging a cell phone, 5 volt usb boost converters get less efficient & provide less current to your phone as battery voltage drops.
Panasonic "3400mah" & Sanyo 2600mah. HKJ's charts.
/Intl-Outdoor%20UR18650FM%202600mAh%20(Black)-Capacity.png)
Ahh I see! Well, it seems convoluted to order Efest IMR (I have sent them an email for a quote), so I may get some Sanyo from FastTech.
Ahh, thank you! I hadn’t settled on the Panasonics. I need to do more reading. >.<
Aww, these are temporarily sold out and apparently discontinued (can’t find anything directly from Panasonic about this though):
http://www.fasttech.com/products/0/10001980/1141101-panasonic-cgr18650ch-rechargeable-2250mah-3-7v
I did find this, which might be biased? I don’t know what kind of Li-ion cells it refers to:
Looks like I won’t be needing that rig after all. There’s the dead short test (“akin to dropping a wrench across the positive and negative terminals”), writ large! But really,
Smoke also exited the vent hole but a fire did not result
You really can be a buzzkill. ![]()
Seriously, Thanks for taking that off my plate!
But he starts with the “Don’t leave the battery in the charger once it is fully charged” method. Somewhere I found some recommendation that Life Cycle would be extended by holding them at “Full Charge” (whether that’s 3.9 or 4.2 — apparently 3.9v Full is another way of extending its Life)… I don’t want to start duelling with PDFs, but I’m working my way through Sony’s Lithium Ion Rechargeable Batteries Technical Handbook but it’s dense. I did find this:
The battery is charged at a constant current to 1CmA to a terminal voltage of 4.2 V under constant-voltage, constat-current [sic] conditions, and after the terminal voltage has reached 4.2V the battery is continuously charged for 28 days at constant voltage
where the UUT passed. That’s consistent with my limited experience with “giveaway” chargers. Jump to the Abuse Tests on p22… I might get an “Intrinsically Safe” rating after all, if this can be backed up empirically.
That’s all just Sony’s opinion, of course, but they seem to have developed some useful information. I’m still looking for ANY analysis of the outgassing in the case of overtemp/overvoltage. Your guy said
Avoid designing airtight battery compartments. In some cases, gases (oxygen, hydrogen) may be given off, and there is a danger of a battery bursting or rupturing if ignited by sparks.
, which is still not “user-friendly”.
3–2 Safety mechanisms
For example, cylindrical batteries are incorporated such safety mechanisms as circuit-breaker devices, safety vents, and PTC devices. The maximum charging voltage of the dedicated recharger for these batteries is set to 4.2 V, but should the recharger malfunction and the maximum setting become invalid, the Li2CO3 added to the cathode would dissociate from around 5 V, and the gas generated as a result would cause an increase in the internal battery pressure. This change in pressure would cause the safety vent to be deformed, removing the cathode lead and cutting off the charging current, thereby ensuring safety. In addition, should the battery be exposed to a fire or some other extreme temperature condition causing a sudden increase in internal pressure, the safety vent would split, averting possible danger.
Moreover, should erroneous use result in the flow of large currents and battery charging and discharging, the PTC device will act to reduce the current
They seem awfully confident, for not disclosing the reactions of the Li2CO3
…
Of course that’s just one source, but you mentioned you’re tired. I figured this would knock you right out. ![]()
Now I’m going to take the easy route & put on my Indiana Jones hat & try to go dig up where the horror stories come from (Actually I think you hit it already: Lithium is not Lithium-Ion!)…
Evermore Dim
Seriously, Thanks for taking that off my plate!
I don’t understand! :~ Did you want to do these tests?
Of course that’s just one source, but you mentioned you’re tired. I figured this would knock you right out.
These look good, no? Nice and shiny… I hope they reply soon! :bigsmile:
It can be annoying to get efest batteries. E-cig sites carry them. Myself & others have done FT product requests.
I don’t normally like getting batteries from ebay but I went for efest imr 10440 from Saftymind (100%, 4,761 reviews)… No efest imr 18650 2000mah listed from saftymind though. :~