Gords pointed out this could be useful so here it is.
Proper Use of 18650s
The proper use of li-ion batteries is a quite complex issue, and one of the most important as misuse of them can lead to an explosion.
What is a Li-ion Battery?
Lithium Ion batteries are very popular in flashlights for a number of reasons. One reason is that the voltage of the battery is very close to the voltage required by most LEDs. This makes it much easier to have them run an LED. They also hold a large amount of power in a small space, they are much more energy dense than Alkaline batteries. This is because unlike alkaline batteries, lithium batteries hold almost as much power at a 3 amp draw as at a 1 amp draw. The other advantage is they are rechargeable, this means we can use our flashlights as much as we want since they simply have to be recharged.
There are 2 main types of Lithium Ion batteries, the first and most common is the LiCo cell. (Lithium Cobalt) these are the cells used in laptop batteries along with many power tool batteries. They have a nominal voltage of 3.7 volts and are 4.2 volts when fully charged. Most of them can be run down to 3 volts, before you should stop using them and recharge them. They can generally supply around 5 amps maximum.
Another type of battery that is very similar to the LiCo cell is the LiMn cell or what is know as an IMR cell. LiMn cells can supply over 20 amps so are great for high current situations. Naturally, something that can supply so much power is quite dangerous if shorted. In some cases they are safer though since they do not catch on fire or explode violently if punctured or crushed.
Both LiMn and LiCo cells have the same voltage, but the LiCo cells tend to have a higher capacity than LiMn cells. Since their voltage varies a large amount during discharge the discharge curve will look like this.

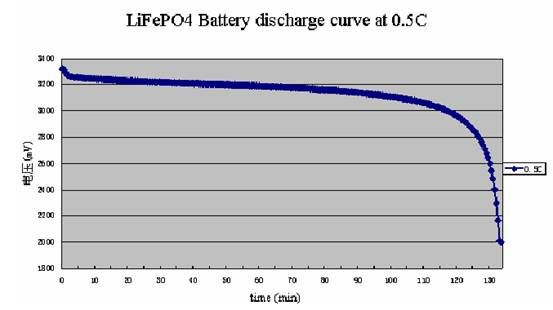
The other main type of Li-ion battery is LiFePo4. Unlike LiCo batteries they have a lower voltage. They have a nominal voltage of 3.2 volts and are 3.5 fully charged. They are considered “dead” when they reach 3 volts. It is because of this that they have a very flat discharge curve. (See Below) They are also much safer, unlike a LiCo cell it is very hard to make them explode or react violently. Because of this LiFePo4 cells are not made with protection circuits. The disadvantage to LiFePo4 batteries is twofold. First, they have a lower capacity. Second, they have a lower voltage and
thus cannot be used to direct drive an LED. This means that some form of circuitry, a boost circuit, is needed in between a LiFePo4 battery and the LED.
Construction of a Li-ion Battery
I'm not going into the chemistry of a li-ion battery, this is simply a explanation of what the different parts are.
So you get something like this in the mail, a protected 18650.

This protected battery is composed of a 18650 battery and a protection circuit.
 (HKJ's picture)
(HKJ's picture)
The protection circuit placed at the negative side of the cell, but is attached to the positive side through a piece of wire. The purpose of the protection circuit is to prevent the battery from being over-discharged (discharged below 3 volts generally), over-charged (charged to above 4.2 volts), or over-current (letting out more than X amps, where X is a value generally around 6).
Then there is the battery itself.

It is composed of a number of different components stacked inside a metal casing which is then wrapped with shrink wrap. This is a before assembly picture.
That green roll is the part that actually holds the energy. It is made up of a positive electrode (a metal oxide), a negative electrode (carbon), and an insulator rolled up. All of these sit in the electrolyte (non-liquid).
Here a diagram of that is.

Now what about the other parts? Here a crappy paint diagram of the top of the battery is.

Ok, so the purpose of the insulator is to prevent the body of the cell (negative) from shorting to the top (positive). The PTC is a positive temperature coefficient. As it heats up the resistance increases, this is a kind of added protection as when the cell is shorted it will heat up. Then as the resistance increases the short will stop. The vent holes allow the battery to vent any gasses, rather than having pressure build up inside the casing of the battery.
There are 3 main types of positive contacts used on the batteries. First, and most common is flat top; these are a simple metal contact no higher than the surrounding label. This is the way batteries generally come from the manufacturer. (Does not apply to NCR18650 series) Then there is raised top, or button top, this has the same contact size as the previous one, but the whole surface is raised. This is needed in lights where the batteries are stacked to ensure contact. Then there is nipple top, this has a small raised top; much smaller. Some lights that have mechanical reverse-polarity protection require nipple tops.
People often use small round magnets to create a a nipple top from a flat top. To do this one simply places a magnet on the top of the battery, this does come with a risk. The magnet can easily slide out of space and short out the battery, it is for this reason people often place a dab of superglue under the magnet. An example of one of these magnets is this.
How to treat LiCo/LiMn Cell in Flashlights
LiCo/LiMn cells are the most dangerous kinds of Li-ion battery. They have and can explode if not treated with respect and care.
First of all, when using them in multi-cell lights make sure they are the same voltage. This means you will need to have a DMM and check the voltage before you load them in the flashlight. This is important because as they discharge if one is at 3.6 volts and the other is at 0 then the one at 0 can get reverse charged. This is how they explode.
Continuing in the mission to prevent reverse charging, one must use cells of the same capacity. This means all of the cells must be identical, and matched to each other. A few notes about this: xxxxfire cells are not considered identical and a cell that has been used a lot is not identical to an unused cell.
How to treat a LiCo/LiMn Cell When Charging
When you charge them you should always use a good charger. Do not cheap out and use a 2 dollar charger from China. Overcharging can also lead to an explosion, there are documented cases where it has.
Lico/LiMn batteries have a complicated way in which they must be charged. For this reason do not try to make your own charger. These are the directions for how Panasonic says to charge them.

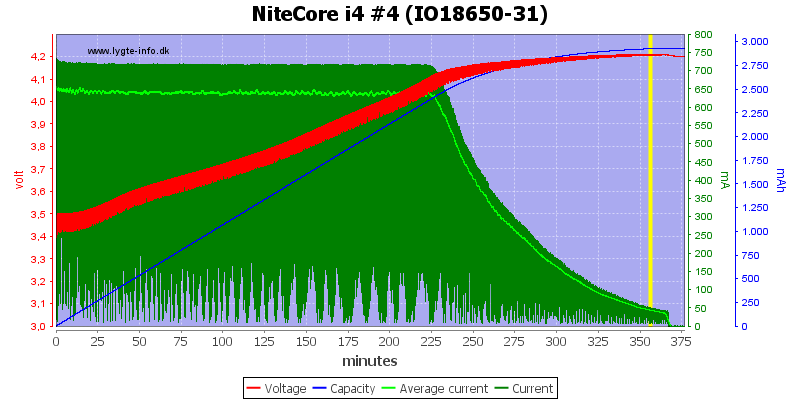
As you can tell this is extremely complicated. As far as I know, there are not any chargers that follow this exactly but there are many that do do a CC/CV curve which is what matters. A CC/CV curve means that first the charger charges the battery with a constant current and lets the voltage rise. Then it switches over to constant voltage and lets the current drop. Here an example of a correct charge curve is. (HKJ)
HKJ has reviews a number of chargers so you can find one that he has approved.
My personal favorite is the Intellicharge I4 which can charge both LiMn/Lico cells and Nimh cells. It is priced very reasonably at 20-25 dollars. They also make an Intellicharge I2 which is equally capable.
Xtar also makes a number of great chargers, all of which follow a CC/CV curve.
So once you have picket out a suitable charger and need to charge your batteries you simply place them inside the charger and wait. One should stay near the batteries while they are charging, and if they start to heat up or smell unplug it immediately.
How to Treat LiFePo4 Cells in Flashlights
LiFePo4 cells are much safer than LiCo/LiMn cells, but the same rules still apply. For ease of use I repeated the information below.
First of all, when using them in multi-cell lights make sure they are the same voltage. This means you will need to have a DMM and check the voltage before you load them in the flashlight. This is important because as they discharge if one is at 3.4 volts and the other is at 0 then the one at 0 can get reverse charged. This is what often causes them to explode.
Continuing in the mission to prevent reverse charging, one must use cells of the same capacity. This means all of the cells must be identical, and matched to each other. A few notes about this: xxxxfire cells are not considered identical and a cell that has been used a lot is not identical to an unused cell.
Also, do not mix LiFePo4 cells with LiCo/LiMn cells.
How to treat a LiFePo4 Cell When Charging
Like LiCo/LiMn cells, LiFePo4 cells should be watched while in the charger.
They also have to be charged with a CC/CV curve, but the difference is they terminate at 3.6 volts.
Currently, if you want a LiFePo4 18650 charger you have to use either a hobby charger or a untested chinese charger. Neither of these is optimal.
However for 16340 LiFePo4 cells, the Xtar MP2 is capable of charging them if set to "3.0V".
One last note about LiFePo4 cells, I have personally tortured them by overcharging, shorting, overdischarging and extremely high discharge rates. Not once have they ever vented.
Estimating Remaining Capacity in Li-ion Batteries
When you measure the resting voltage of a lithium ion battery you can get a rough idea of how much capacity is left in it. Use this table.

Choosing The Right Battery
First of all, any battery that has "fire" in it's name is not suitable for use in multi-cell lights. This is because even if they may be rated at the same capacity, they are most likely not equal and could become unbalanced under high loads.
For 18650 go to post 1, for anything smaller than a 18650 go to post 2. For anything bigger go to post 3.
Feel free to comment and correct me on any mistakes.

/Intl-Outdoor%20UR18650FM%202600mAh%20(Black)-Capacity.png)
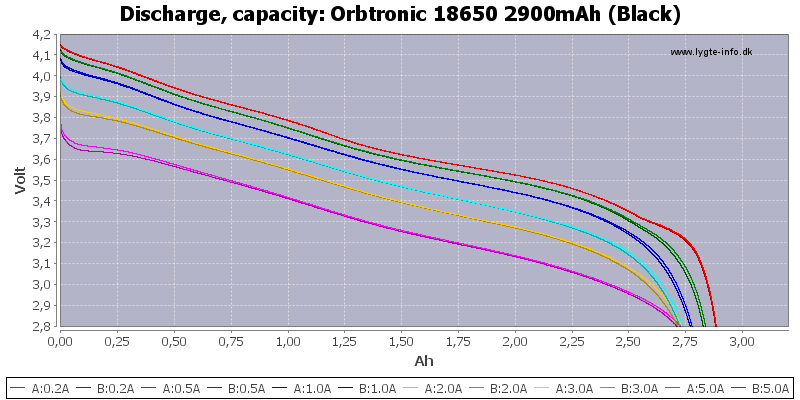
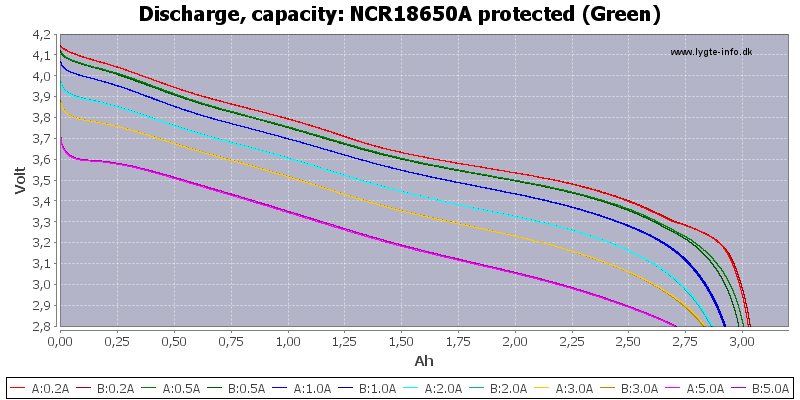
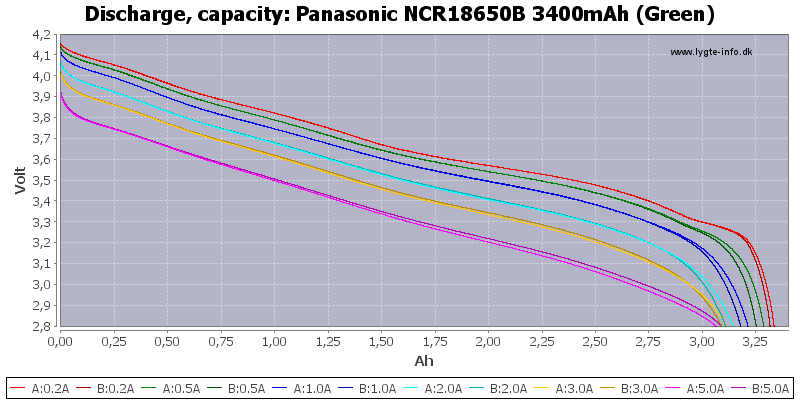
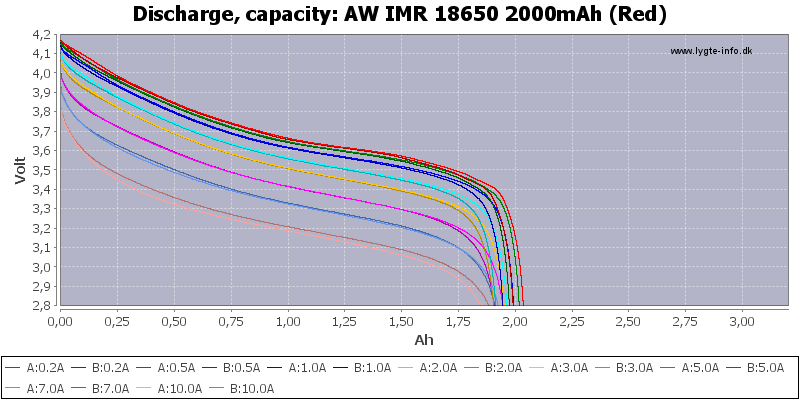
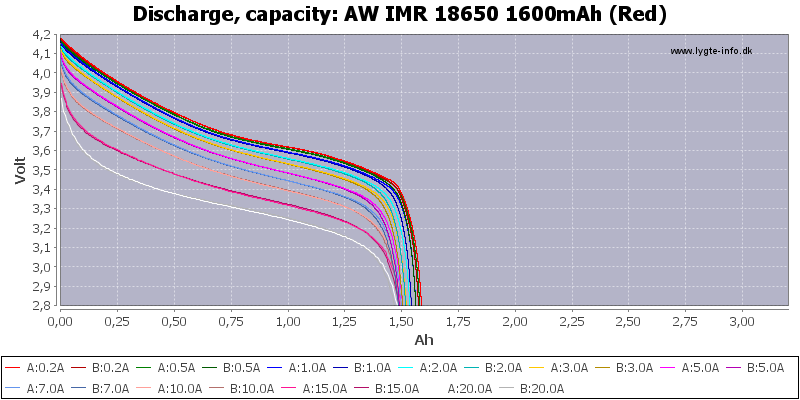
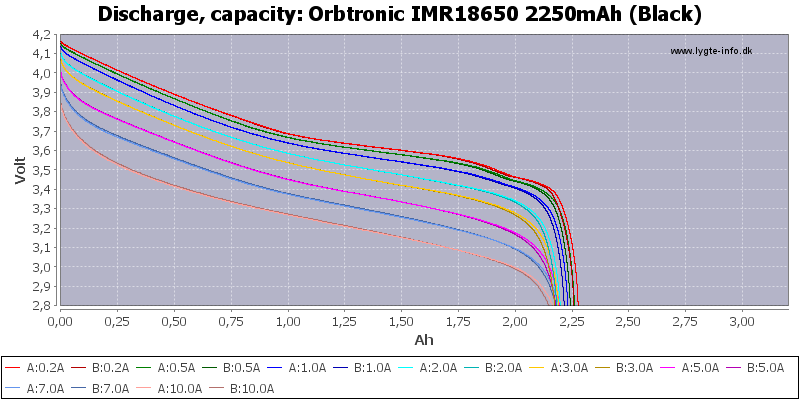
 (Not HKJ)
(Not HKJ)
/AW%20IMR%2016340%20550mAh%20(Red)-Capacity.png)
/AW%2016340%20ICR123%20750mAh%20(Black)-Capacity.png)
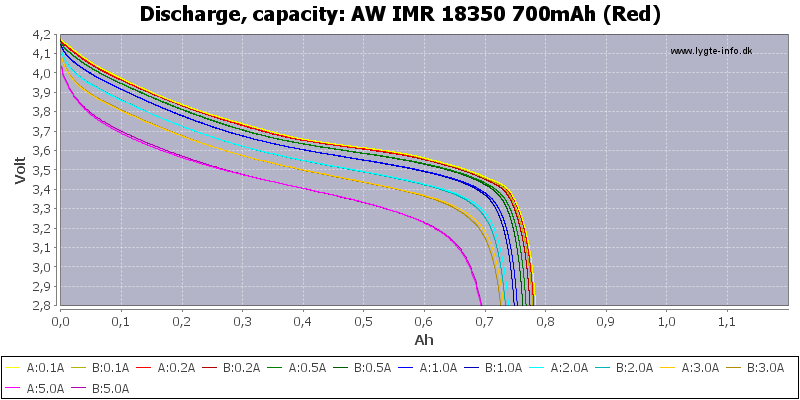
/AW%20IMR%2014500%20600mAh%20(Red)-Capacity.png)
/AW%2014500%20750mA%20(Black)-Capacity.png)
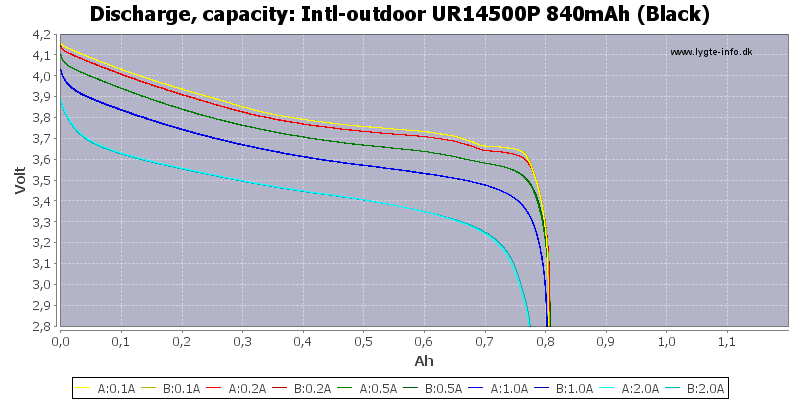
/AW%20ICR10440%20350mAh%20(Blue)-Capacity.png)
/Efest%20IMR10440%20V1%20350mAh%20(Black)-Capacity.png)
%204.3V/Keeppower%2016650%202000mAh%20(Black)%204.3V-Capacity.png)
/Intl-outdoor%20Sanyo%2016650%202100mAh%20(Red)-Capacity.png)
/EagleTac%2017650%201600mAh%20(Black)-Capacity.png)
/Keeppower%2018500%201500mAh%20(Black)-Capacity.png)
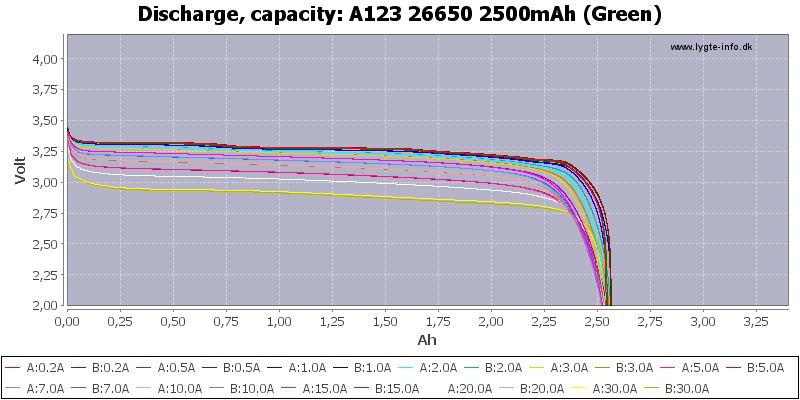
/Keeppower%2026650%204000mAh%20(Black)-Capacity.png)
/TrustFire%20TF26650%205000mAh%20(Flame)-Capacity.png)
/King%20Kong%20ICR26650%204000mAh%20(Gold)-Capacity.png)
/King%20Kong%20INR26650E%204000mAh%20(Gold)-Capacity.png)
/UltraFire%20BRC32600%204000mAh%203.0V%20(Green-white)-Capacity.png)